
Comparison of Structural Features of Humins Formed Catalytically from Glucose, Fructose, and 5-Hydroxymethylfurfuraldehyde | Energy & Fuels

Gd(III) and Yb(III) Complexes Derived from a New Water-Soluble Dioxopolyazacyclohexane Macrocycle | ACS Omega

Lanthanide(III) Complexes with Ligands Derived from a Cyclen Framework Containing Pyridinecarboxylate Pendants. The Effect of Steric Hindrance on the Hydration Number | Inorganic Chemistry

Gd(III) and Yb(III) Complexes Derived from a New Water-Soluble Dioxopolyazacyclohexane Macrocycle | ACS Omega

The chemical consequences of the gradual decrease of the ionic radius along the Ln-series - ScienceDirect

Picolinate-Containing Macrocyclic Mn2+ Complexes as Potential MRI Contrast Agents | Inorganic Chemistry

Gd(III) and Yb(III) Complexes Derived from a New Water-Soluble Dioxopolyazacyclohexane Macrocycle | ACS Omega

Macromolecules, Dendrimers, and Nanomaterials in Magnetic Resonance Imaging: The Interplay between Size, Function, and Pharmacokinetics | Chemical Reviews
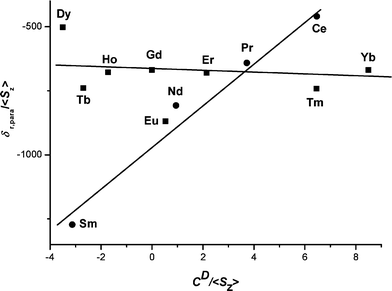
The structure of the lanthanide aquo ions in solution as studied by 17O NMR spectroscopy and DFT calculations - Dalton Transactions (RSC Publishing) DOI:10.1039/B714801A
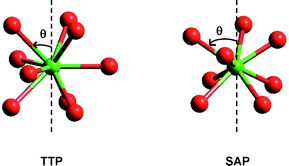
The structure of the lanthanide aquo ions in solution as studied by 17O NMR spectroscopy and DFT calculations - Dalton Transactions (RSC Publishing) DOI:10.1039/B714801A

1H and 17O NMR Relaxometric and Computational Study on Macrocyclic Mn(II) Complexes | Inorganic Chemistry

Lanthanide(III) Complexes with Ligands Derived from a Cyclen Framework Containing Pyridinecarboxylate Pendants. The Effect of Steric Hindrance on the Hydration Number | Inorganic Chemistry

Gd(III) and Yb(III) Complexes Derived from a New Water-Soluble Dioxopolyazacyclohexane Macrocycle | ACS Omega

PDF) Americium(III) and europium(III) complex formation with lactate at elevated temperatures studied by spectroscopy and quantum chemical calculations
![General Acid-Type Catalysis in the Dehydrative Aromatization of Furans to Aromatics in H-[Al]-BEA, H-[Fe]-BEA, H-[Ga]-BEA, and H-[B]-BEA Zeolites | The Journal of Physical Chemistry C General Acid-Type Catalysis in the Dehydrative Aromatization of Furans to Aromatics in H-[Al]-BEA, H-[Fe]-BEA, H-[Ga]-BEA, and H-[B]-BEA Zeolites | The Journal of Physical Chemistry C](https://pubs.acs.org/cms/10.1021/acs.jpcc.7b02344/asset/images/large/jp-2017-02344m_0001.jpeg)
General Acid-Type Catalysis in the Dehydrative Aromatization of Furans to Aromatics in H-[Al]-BEA, H-[Fe]-BEA, H-[Ga]-BEA, and H-[B]-BEA Zeolites | The Journal of Physical Chemistry C




