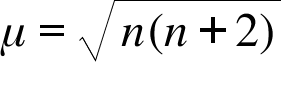ReasonThe spin only magnetic moment of an ion is equal to sqrt {n(n+2)} where n is the number of unpaired electrons in the ion.AssertionThe spin only magnetic moment of Sc^{3+} is 1.73

The highest value of the calculated spin-only magnetic moment (in BM) among all the transition - YouTube







![The magnetic moment (spin only) of [NiCl4]2– is - askIITians The magnetic moment (spin only) of [NiCl4]2– is - askIITians](https://files.askiitians.com/cdn1/cms-content/common/www.askiitians.comonlinetestforumsimages204-1316_sataug1615-54-35.jpg.jpg)


![Assamese] What is 'spin only' formula? Assamese] What is 'spin only' formula?](https://d10lpgp6xz60nq.cloudfront.net/ss/web-overlay-thumb/8950532.webp)
![The spin-only magnetic moments of k\u0104[fe(oxalate),] and k,[ru(oxalate),] are The spin-only magnetic moments of k\u0104[fe(oxalate),] and k,[ru(oxalate),] are](https://cdn.eduncle.com/library/scoop-files/2020/7/image_1594373088294.jpg)